Medical College of Wisconsin Multiple Sclerosis & Neuroimmunology Research
Researchers in Neurology, led by Ahmed Obeidat, MD, PhD, investigate a variety of neuroimmunologic disorders, including multiple sclerosis, neuromyelitis optica, and autoimmune encephalitis. Their goals are to understand the etiology and pathophysiology of these disorders, improve diagnosis through advanced imaging and quantitative neurological testing, and develop and evaluate new treatments.
INTERTWINE Lab
ImmuNology, Tool dEvelopment, Risk Factors, Treatment Safety and Wellbeing In MS and other NEuroimmune disorders
The INTERTWINE lab, headed by Ahmed Obeidat, MD, PhD, includes a team of clinicians, scientists, fellows, residents and students working together to investigate timely issues related to multiple sclerosis (MS) and other neuroimmune disorders. The etiology of MS remains poorly understood. Our lab examines hypotheses related to the role of environmental factors, including viruses in MS development. We further extend our investigations to other neuroimmune disorders, including Neuromyelitis Optica spectrum disorders and autoimmune encephalitis. We use clinical assays while aiming to pursue more intricate assessments of viral roles in the development of neuroimmune disorders. Our team performs detailed immunophenotyping to advance our understanding of changes in the adaptive immune system in MS before and after treatment with disease modifying therapies. We aim to further characterize response to treatment and predict long-term safety concerns, if any.
We also realize the need for a sensitive tool to assess abilities in MS and related disorders. Our team goal is to help develop a user-friendly and sensitive tool benefiting from current technological advancements in the field of biomechanical engineering. We are currently funded by the center for immunology at MCW and also have a pilot grant from the MCW Research Affairs Committee. Our team is active in writing grant proposals to fund our continuing research endeavors.
Projects
Clinical Trials
Traditional Versus Early Aggressive Therapy for Multiple Sclerosis Trial (TREAT-MS)
Sponsor: John Hopkins University – Funding: Patient-Centered Outcomes Research Institute (PCORI) and National MS Society
PI: Ahmed Obeidat, MD, PhD
Status: Active – recruiting
A Phase III Multicenter, Randomized, Double-Blind, Double-Dummy, Parallel-Group Study to Evaluate the Efficacy and Safety of Fenebrutinib Compared with Ocrelizumab in Adult Patients with Primary Progressive Multiple Sclerosis
Sponsor: Genentech
PI: Ahmed Obeidat, MD, PhD
Status: Active – recruiting
A Multicenter, Longitudinal, Open-Label, Single-Arm Study Describing Cognitive Processing Speed Changes In Relapsing Multiple Sclerosis Subjects Treated With Ozanimod (RPC-1063)
Sponsor: Celgene
PI: Ahmed Obeidat, MD, PhD
Status: Active – recruiting
Prospective Study to Assess Disease Activity and Biomarkers in Minority Participants With Relapsing Multiple Sclerosis (RMS) After Initiation and During Treatment With Ocrelizumab
Sponsor: Genentech
PI: Ahmed Obeidat, MD, PhD
Status: Active – recruiting
Exploring the safety and tolerability of conversion from oral or injectable disease-modifying therapies to dose titrated Oral Siponimod in patients with advancing forms of relapsing multiple sclerosis: A 6-month open label, multicenter Phase IIIb study (EXCHANGE)
Sponsor: Novartis
PI: Ahmed Obeidat, MD, PhD
Status: Active – recruiting
Additional Information
Translating Oral Immunoactive Proteins into Therapy for Autoimmune Diseases
 Experimental autoimmune encephalitis (EAE) is an inflammatory autoimmune process of the CNS that resembles multiple sclerosis (MS) and provides a useful animal model for the evaluation of mechanisms of action for potential immunomodulatory therapies. Oral ACTH can decrease clinical disease, IL-17, and Th1-like encephalitogenic IFN-g secretion, and increase Treg frequency. The mechanism by which oral ACTH decreases inflammatory proteins and increases Treg cell frequencies is unknown. IL-6 is a pivotal cytokine in the gut that determines the relative frequencies of Th17 vs Treg cells. In collaboration with Dr. Bonnie Dittel’s laboratory at the Blood Research Institute (Versiti), Dr. Brod and his team examined whether oral ACTH inhibits IL-6 in gut associated lymphoid tissue (GALT) in EAE. B6 mice were immunized with MOG peptide 35-55 and gavaged with scrambled ACTH (s-MSH) peptide or ACTH 1-39 during ongoing disease.
Experimental autoimmune encephalitis (EAE) is an inflammatory autoimmune process of the CNS that resembles multiple sclerosis (MS) and provides a useful animal model for the evaluation of mechanisms of action for potential immunomodulatory therapies. Oral ACTH can decrease clinical disease, IL-17, and Th1-like encephalitogenic IFN-g secretion, and increase Treg frequency. The mechanism by which oral ACTH decreases inflammatory proteins and increases Treg cell frequencies is unknown. IL-6 is a pivotal cytokine in the gut that determines the relative frequencies of Th17 vs Treg cells. In collaboration with Dr. Bonnie Dittel’s laboratory at the Blood Research Institute (Versiti), Dr. Brod and his team examined whether oral ACTH inhibits IL-6 in gut associated lymphoid tissue (GALT) in EAE. B6 mice were immunized with MOG peptide 35-55 and gavaged with scrambled ACTH (s-MSH) peptide or ACTH 1-39 during ongoing disease.
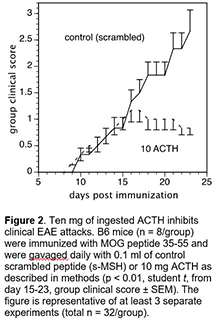
Ingested (oral) ACTH inhibited ongoing clinical disease, as shown in Figure 2. In the lamina propria (LP) immune compartment, there were significantly fewer CD11b+ and CD4+ IL-6 producing lymphocytes from ACTH fed mice compared to s-MSH fed mice. There was also a decrease in the frequency of IL-17 and IFN-g producing spleen cells and an increase in CD4+FoxP3+ Treg cell frequency in ACTH fed mice compared to s-MSH fed control spleens.
The results demonstrate that ingested ACTH inhibits EAE clinical disease by inhibiting IL-6 in the GALT. The administration of endogenous immune-active proteins via the gut offers an alternative to systemic application with ease of administration in chronic clinical use (Rollwagen and Baqar, 1996) and patient convenience, and would be a great step forward because gut delivery is easy, well tolerated, and inexpensive with a favorable therapeutic index. We have already administered ACTH to humans but lacked information on the mechanism of ACTH in the gut. These experiments in mice help us understand the mechanism of action and provide a rationale for pursuing further clinical trials are planned in MS subjects.
Figure 3
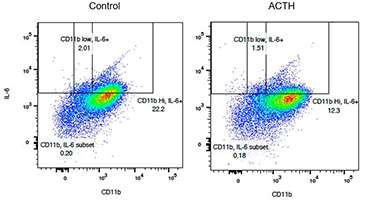
In addition, our studies have shown that IL-6, a critical cytokine in gut immune cells, is inhibited by oral ACTH. The flow diagrams in Figure 3 above show that oral ACTH decreases the frequency of IL-6 producing CD11b+ dendritic cells in the lamina propria from 22% to 12%.
Figure 4
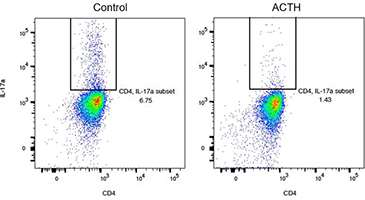
Next we showed that IL-17, a critical cytokine in EAE generation is also inhibited by oral ACTH in the lamina propria. The flow diagrams in Figure 4 above show that oral ACTH decreases the frequency of IL-6 producing CD11b+ dendritic cells in the lamina propria from 6.75% to 1.43%.
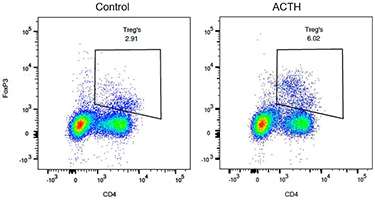
Figure 5
Next we showed that regulatory T cells (Tregs) that diminish disease in MS, are increased by oral ACTH in the spleen. The flow diagrams in Figure 5 above show that oral ACTH increases the frequency of Tregs cells in the lamina propria from 2.9% to 6.0%.
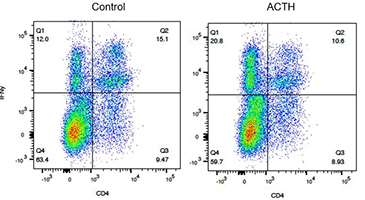
Figure 6
Finally, as shown above in Figure 6 above, oral ACTH decreases the frequency of IFN-g producing T cells in the CNS (central nervous system) of the mice fed ACTH in contrast to the mice fed control peptide.
These experiments showing that IL-6, other inflammatory factors, and clinical disease can be controlled by ingesting an immune-active protein will lead to human clinical trials using oral ACTH in MS.
Meet Our Team
Faculty

Ahmed Zayed Obeidat, MD, PhD
Associate Professor
Bonnie N. Dittel, PhD
Senior Investigator, Versiti Blood Research Institute; Professor, Microbiology & Immunology

Jessica Francis, MD
Associate Professor

Sam I. Hooshmand, DO
Assistant Professor

Andrew P. Klein, MD
Chief, Associate Professor; Associate Program Director
Aristide Merola, MD, PhD
Neurologist and Movement Disorder Specialist, Ohio State University

L. Tugan Muftuler, PhD
Professor

Vera Tarakanova, PhD
Professor
Staff and Students
Calin Dumitrescu
Medical Student
Leah Hoffman
Pharmacy Student, Concordia University
Samuel Marcucci
Medical Student, University of Cincinnati