
John E. Baker, PhD
Professor, Surgery (Pediatrics); Secondary Faculty
Locations
- Biochemistry
MFRC 4051
Contact Information
General Interests
Education
Biography
Research Experience
- Cardiovascular Agents
- Cardiovascular Diseases
- Cardiovascular Physiological Phenomena
- Cardiovascular System
- Microbiota
- radiation effects
- Radiation Effects
- Radiation Genetics
- Radiation Injuries
- Radiation Injuries, Experimental
- Radiation Tolerance
- Radiation, Ionizing
Clinical Expertise
- Clinical Trial
- Clinical Trials, Phase II as Topic
- Drugs, Investigational
Leadership Positions
- Chair, Institutional Animal Care and Use Committee
Research Interests
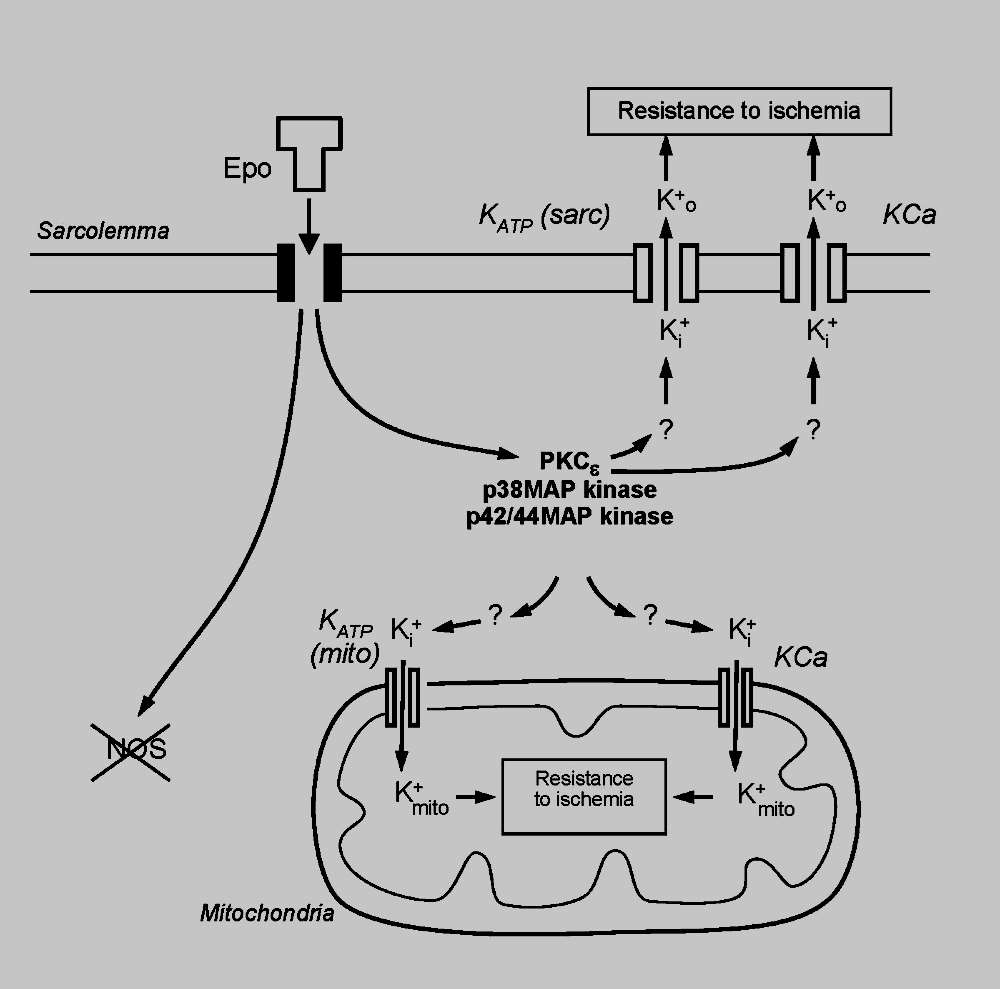 To determine a possible role for erythropoietin in cardioprotection during surgical ischemia and the underlying mechanisms we treated infant rabbit hearts with human recombinant erythropoietin prior to ischemia. The objectives of our study were to determine whether exposure of the heart to erythropoietin would immediately increase its resistance to subsequent ischemia, the erythropoietin concentration that confers optimal protection of the heart, the involvement and cellular location of protein kinase signaling pathways, and the role of potassium channels and nitric oxide synthase in mediating cardioprotection. Our study shows that erythropoietin exerts a concentration-and time-dependent cardioprotective effect. The mechanisms underlying erythropoietin-induced cardioprotection involves activation of PKC-epsilon, p38 MAP kinase and p42/44 MAP kinase with increased resistance to myocardial ischemia mediated by potassium channels but not by nitric oxide synthase. The optimal concentration of 1.0 U/ml needed to confer protection against cardiac ischemia is approximately 100 times above levels present during chronic hypoxia and 500 times above erythropoietin levels present in the circulation of normoxic rabbits. Increased resistance to myocardial ischemia is observed immediately after treatment with erythropoietin, indicating that induction of new genes is not necessary for its cardioprotective effect to be manifested. We believe our study is the first to demonstrate the biological effects of erythropoietin are mediated by a signal pathway that results in immediate activation of two potassium channels, the KATP and the KCa channel. These studies prompted us to hypothesize that erythropoietin would also be able to protect the infant human heart against injury during surgical repair of congenital heart disease. We plan to conduct a clinical trial to test this hypothesis in the setting of the cardiac operating room at Children's Wisconsin.
To determine a possible role for erythropoietin in cardioprotection during surgical ischemia and the underlying mechanisms we treated infant rabbit hearts with human recombinant erythropoietin prior to ischemia. The objectives of our study were to determine whether exposure of the heart to erythropoietin would immediately increase its resistance to subsequent ischemia, the erythropoietin concentration that confers optimal protection of the heart, the involvement and cellular location of protein kinase signaling pathways, and the role of potassium channels and nitric oxide synthase in mediating cardioprotection. Our study shows that erythropoietin exerts a concentration-and time-dependent cardioprotective effect. The mechanisms underlying erythropoietin-induced cardioprotection involves activation of PKC-epsilon, p38 MAP kinase and p42/44 MAP kinase with increased resistance to myocardial ischemia mediated by potassium channels but not by nitric oxide synthase. The optimal concentration of 1.0 U/ml needed to confer protection against cardiac ischemia is approximately 100 times above levels present during chronic hypoxia and 500 times above erythropoietin levels present in the circulation of normoxic rabbits. Increased resistance to myocardial ischemia is observed immediately after treatment with erythropoietin, indicating that induction of new genes is not necessary for its cardioprotective effect to be manifested. We believe our study is the first to demonstrate the biological effects of erythropoietin are mediated by a signal pathway that results in immediate activation of two potassium channels, the KATP and the KCa channel. These studies prompted us to hypothesize that erythropoietin would also be able to protect the infant human heart against injury during surgical repair of congenital heart disease. We plan to conduct a clinical trial to test this hypothesis in the setting of the cardiac operating room at Children's Wisconsin.
Publications
-
Advancing cell-free DNA as a biomarker of damage to heart caused by ionizing radiation.
(Wallisch E, Tomita-Mitchell A, Liang HL, Szabo A, Lenarczyk M, Kwitek A, Smith JR, Tutaj M, Baker JE.) J Radiat Res. 2025 May 23;66(3):329-340 PMID: 40304705 PMCID: PMC12100473 SCOPUS ID: 2-s2.0-105006519967 04/30/2025
-
(Naas CJ, Saleh HO, Engel TW 2nd, Gutterman DD, Szabo A, Grawey T, Weston BW, Monti CE, Baker JE, Labinski J, Tang L, Jasti J, Bartos JA, Kalra R, Yannopoulos D, Riccardo Colella M, Aufderheide TP.) Resuscitation. 2025 Apr;209:110567 PMID: 40057016 SCOPUS ID: 2-s2.0-105000556919 03/09/2025
-
Radiation-Induced Cardiotoxicity in Hypertensive Salt-Sensitive Rats: A Feasibility Study
(An D, Kriegel A, Kumar S, Himburg H, Fish B, Klawikowski S, Rowe D, Lenarczyk M, Baker J, Ibrahim ES.) Life. June 2025;15(6) SCOPUS ID: 2-s2.0-105009049617 06/01/2025
-
(Raber J, Chaudhari M, De la Torre A, Holden S, Kessler K, Glaeser B, Lenarczyk M, Leonard SW, Borg A, Kwok A, Patel C, Kronenberg A, Olsen CM, Willey JS, Morré J, Choi J, Stevens JF, Bobe G, Minnier J, Baker J.) Front Physiol. 2024;15:1486767 PMID: 39605860 PMCID: PMC11598337 SCOPUS ID: 2-s2.0-85210519824 11/28/2024
-
(Lenarczyk M, Alsheikh AJ, Cohen EP, Schaue D, Kronenberg A, Geurts A, Klawikowski S, Mattson D, Baker JE.) Toxics. 2023 Feb 16;11(2) PMID: 36851074 PMCID: PMC9959763 SCOPUS ID: 2-s2.0-85149176377 03/01/2023
-
(Raber J, Holden S, Kessler K, Glaeser B, McQuesten C, Chaudhari M, Stenzel F, Lenarczyk M, Leonard SW, Morré J, Choi J, Kronenberg A, Borg A, Kwok A, Stevens JF, Olsen C, Willey JS, Bobe G, Minnier J, Baker JE.) Front Physiol. 2023;14:1316186 PMID: 38260101 PMCID: PMC10800373 01/23/2024
-
(Lenarczyk M, Kronenberg A, Mäder M, Komorowski R, Hopewell JW, Baker JE.) PLoS One. 2023;18(4):e0283877 PMID: 37099482 PMCID: PMC10132632 SCOPUS ID: 2-s2.0-85153985230 04/26/2023
-
(Ibrahim EH, Sosa A, Brown SA, An D, Klawikowski S, Baker J, Bergom C.) Tomography. 2022 Dec 22;9(1):36-49 PMID: 36648991 PMCID: PMC9844312 SCOPUS ID: 2-s2.0-85146484422 01/18/2023
-
(Lenarczyk M, Alsheikh AJ, Cohen EP, Schaue D, Kronenberg A, Geurts A, Klawikowski S, Mattson D, Baker JE.) Toxics. 2022 Dec 18;10(12) PMID: 36548630 PMCID: PMC9783591 12/23/2022
-
(Aufderheide TP, Engel TW 2nd, Saleh HO, Gutterman DD, Weston BW, Pepe PE, Baker JE, Labinski J, Debaty G, Tang L, Szabo A, Kalra R, Yannopoulos D, Colella MR.) Resuscitation. 2021 Dec;169:45-52 PMID: 34666124 SCOPUS ID: 2-s2.0-85118490179 10/20/2021
-
(Raber J, Holden S, Sudhakar R, Hall R, Glaeser B, Lenarczyk M, Rockwell K, Nawarawong N, Sterrett J, Perez R, Leonard SW, Morré J, Choi J, Kronenberg A, Borg A, Kwok A, Stevens JF, Olsen CM, Willey JS, Bobe G, Baker J.) Front Physiol. 2021;12:746509 PMID: 34646164 PMCID: PMC8503608 SCOPUS ID: 2-s2.0-85116920048 10/15/2021
-
(Borg AM, Baker JE.) Synth Biol (Oxf). 2021;6(1):ysab023 PMID: 34522784 PMCID: PMC8434797 09/16/2021

