
Christopher M. Olsen, PhD
Professor, Director of Graduate Studies
Locations
- Pharmacology and Toxicology
Contact Information
General Interests
Education
MS, Texas Tech University, 1998
BA, Baylor University, 1994
Research Experience
- Brain Concussion
- Brain Injuries
- Brain Injury, Chronic
- Cocaine-Related Disorders
- Exploratory Behavior
- Motivation
- Opioid-Related Disorders
- Substance-Related Disorders
MCW Program / Core Facilities
- Neuroscience Research Center
Research Interests
Olsen Lab
Addiction is a chronically relapsing disorder that is strongly modulated by positive and negative environmental factors. Our lab (olsenlab.org) focuses on how these environmental factors can influence drug intake and drug seeking using rat and mouse models of addiction.
We have two major projects to investigate environmental factors and addiction:
Environmental modulation of drug seeking ensembles. Transgenic approaches are used to identify and manipulate cells that are engaged during drug seeking following intravenous drug self-administration. We are studying the effects of positive and negative environmental factors on drug seeking and the activity of cells within drug seeking ensembles.
The impact of mild traumatic brain injury (mTBI) on addiction and mesocorticolimbic function. This work is done in collaboration with Drs. Brain Stemper and Matthew Budde in the Department of Biomedical Engineering and Neurosurgery, respectively. We are examining the effects of mTBI on drug self-administration, drug seeking, and mesocorticolimbic function using diffusion tensor imaging (DTI) and functional connectivity.
View the full publication list.
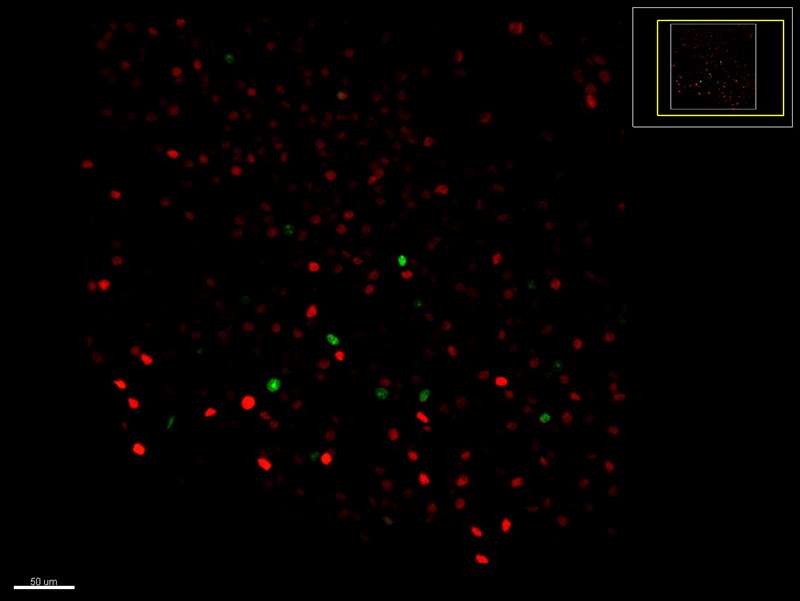
Identification of neuronal ensembles involved in persistent drug seeking. TetTag (green) cells are those engaged during an initial drug seeking session, Fos+ (red) cells are those engaged during a second drug seeking session 2 weeks later.

Quantification of monocytes/microglia. Iba1+ (green) cells are surface rendered for complex analysis to examine long-lasting effect of mild traumatic brain injury on immune cells within the brain. Gray=nuclei (DAPI).
Publications
-
(Everson CA, Szabo A, Olsen CM, Glaeser BL, Raff H.) Sleep. 2025 Sep 09;48(9) PMID: 40253577 PMCID: PMC12417021 SCOPUS ID: 2-s2.0-105015520801 04/20/2025
-
(Liu S, Nawarawong N, Liu X, Liu QS, Olsen CM.) Transl Psychiatry. 2024 Sep 23;14(1):387 PMID: 39313502 PMCID: PMC11420216 SCOPUS ID: 2-s2.0-85204921376 09/24/2024
-
(Sarka BC, Liu S, Banerjee A, Stucky CL, Liu QS, Olsen CM.) Addict Biol. 2024 Aug;29(8):e13430 PMID: 39121884 PMCID: PMC11315577 SCOPUS ID: 2-s2.0-85200756850 08/10/2024
-
(Glaeser B, Panariello V, Banerjee A, Olsen CM.) Drug Alcohol Depend. 2024 Feb 01;255:111077 PMID: 38228055 PMCID: PMC10869844 SCOPUS ID: 2-s2.0-85182578672 01/16/2024
-
(Raber J, Chaudhari M, De la Torre A, Holden S, Kessler K, Glaeser B, Lenarczyk M, Leonard SW, Borg A, Kwok A, Patel C, Kronenberg A, Olsen CM, Willey JS, Morré J, Choi J, Stevens JF, Bobe G, Minnier J, Baker J.) Front Physiol. 2024;15:1486767 PMID: 39605860 PMCID: PMC11598337 SCOPUS ID: 2-s2.0-85210519824 11/28/2024
-
(Olsen CM, Glaeser BL, Szabo A, Raff H, Everson CA.) Physiol Behav. 2023 Dec 01;272:114372 PMID: 37805135 PMCID: PMC10841994 SCOPUS ID: 2-s2.0-85173957688 10/08/2023
-
(Chiariello R, McCarthy C, Glaeser BL, Shah AS, Budde MD, Stemper BD, Olsen CM.) Behav Brain Res. 2023 Feb 13;438:114181 PMID: 36330906 PMCID: PMC9993345 SCOPUS ID: 2-s2.0-85140915448 11/05/2022
-
(Raff H, Glaeser BL, Szabo A, Olsen CM, Everson CA.) Stress. 2023 Jan;26(1):2185864 PMID: 36856367 PMCID: PMC10339708 SCOPUS ID: 2-s2.0-85150079770 03/02/2023
-
(Muelbl MJ, Glaeser BL, Shah AS, Chiariello RA, Nawarawong NN, Stemper BD, Budde MD, Olsen CM.) Addict Biol. 2022 Mar;27(2):e13134 PMID: 35229952 PMCID: PMC8896287 SCOPUS ID: 2-s2.0-85125380733 03/02/2022
-
Editorial: Behavioral outcomes of traumatic brain injury.
(Olsen CM, Herrold AA, Conti AC, Vonder Haar C.) Front Behav Neurosci. 2022;16:1010395 PMID: 36160681 PMCID: PMC9490366 09/27/2022
-
(Stemper BD, Shah A, Chiariello R, McCarthy C, Jessen K, Sarka B, Seifert J, Budde MD, Wang K, Olsen CM, McCrea M.) Front Behav Neurosci. 2022;16:805124 PMID: 35368301 PMCID: PMC8965565 SCOPUS ID: 2-s2.0-85127445249 04/05/2022

