
Sally Twining, PhD
Emeritus Professor
Locations
- Biochemistry
BSB 309
Contact Information
Education
Biography
Research Experience
- Breast Neoplasms
- Corneal Diseases
- Corneal Neovascularization
- Corneal Opacity
- Enzyme Activation
- Keratitis
- Keratoconus
- Maspin
- Pseudomonas aeruginosa
- Receptors, Proteinase-Activated
- Serpins
- Thrombin
Leadership Positions
- Chair M1-2 Subcommittee Curriculum and Evaluation Committee: 2002-2005
- Chair Women's Faculty Council: 2005-2006
- President Faculty Council: 2008-2009
- Sr Vice President Faculty Council: 2007-2008
- Vice President: 2006-2007
Research Interests
The two major projects in the laboratory are 1) elucidation of the role of proteinases and proteinase inhibitors in corneal wound healing and corneal ulceration and 2) exploration of the effects of maspin in corneal wound healing and in carcinogenesis.
The cornea has two major functions; refraction of light and protection of the eye. 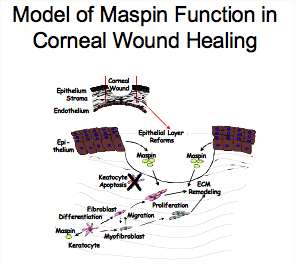 Retention of the avascularity and transparency of the cornea is crucial for vision. Understanding the mechanism of normal and abnormal corneal wound healing is important because of the approximately 1 million refractive surgeries such as LASIK performed each year in the US. Upon wounding, the external barrier of the cornea is reassembled through the movement of the epithelial cells from the non-injured regions of the cornea followed by cell division and stratification of the epithelial layer. In the stroma, the cells near the injury undergo apoptosis. Cells more distant from the wound divide and undergo a phenotypic change to form mobile fibroblasts and with time the formation of contractile myofibroblasts. The fibroblasts and myofibroblasts remodel the stromal area of apoptosis. With time this area is repopulated with the quiescent normal keratocytes. In humans, this process can continue for at least 7-10 years.
Retention of the avascularity and transparency of the cornea is crucial for vision. Understanding the mechanism of normal and abnormal corneal wound healing is important because of the approximately 1 million refractive surgeries such as LASIK performed each year in the US. Upon wounding, the external barrier of the cornea is reassembled through the movement of the epithelial cells from the non-injured regions of the cornea followed by cell division and stratification of the epithelial layer. In the stroma, the cells near the injury undergo apoptosis. Cells more distant from the wound divide and undergo a phenotypic change to form mobile fibroblasts and with time the formation of contractile myofibroblasts. The fibroblasts and myofibroblasts remodel the stromal area of apoptosis. With time this area is repopulated with the quiescent normal keratocytes. In humans, this process can continue for at least 7-10 years.
Our previous work showed that prothrombin, the precursor of the coagulation protease thrombin, is both found within corneal epithelial, stromal and endothelial cells and associated with the extracellular matrix of the stroma. Not only is prothrombin present but also its mRNA. In addition to prothrombin, the message and protein for the other coagulation factors of the intrinsic and extrinsic pathways are found in the cornea. During wound healing, cornea synthesized thrombin, the last protease of the coagulation pathway, is activated by coagulation factors synthesized by the corneal epithelial and stromal cells. Fibrinogen is converted to fibrin and deposited on the surface of the injured cornea and within the stromal matrix. The roles of thrombin, in addition to cleavage of fibrinogen to fibrin, in corneal wound healing are being explored. One of these is cleavage of protease activated receptors, PARs, revealing a cryptic ligand. Upon binding this ligand to the PAR binding site, multiple signaling pathways are activated. These pathways can regulate processes such as cell proliferation, adhesion and migration and can modify gene expression. The effect of thrombin on proteases in the plasminogen activator system, members of the CCN family of proteins, Cyr61 and CTGF, as well as, other growth factors and chemokines are being explored in corneal cells.
One of the major causes of corneal ulceration is bacterial keratitis. Bacteria invasion of host tissues requires proteases that degrade host tissues. Pseudomonas aeruginosa is of specific concern because it colonizes on soft contact lenses. This organism produces a number of secreted factors that alter host functions including the protease Pseudomonas elastase. There are two forms of this enzyme but the functional implications of these differences are not known and are the focus of our studies.
Maspin is a non-inhibitory member of the Serpin (SERpin Protease INhibitor) family which is synthesized by epithelial and endothelial cells and stromal keratocytes in the normal cornea. It stimulates adhesion of cells to extracellular matrix molecules and inhibits migration and invasion of cells including corneal stromal fibroblasts and carcinoma cells. In addition, maspin inhibits angiogenesis though inhibition of vascular endothelial cell proliferation and migration. Our previous work showed that the reactive site loop peptide is sufficient for stimulation of adhesion, inhibition of migration and invasion. Our model of maspin function in the cornea includes regulation of migration of corneal cells through downregulation of maspin synthesis in the stromal fibroblast and myofibroblast phenotypes which allows migration of these cells into the wounded area. Downregulation of maspin synthesis by stromal fibroblasts and myofibroblasts involves DNA and histone methylation in the CpG islands in the promoter. We hypothesize that maspin released by the intact epithelial layer above the wounded cornea inhibits migration of the cells as they repopulate the wounded area. Corneal epithelial cells release maspin through non-classical mechanisms; exosomes and cell death. This released maspin is post-translationally phosphorylated at one or more of eight serine and threonine residues. The functional implications of phosphorylation of maspin is being explored as well as characterization of the locations of phosphorylation on intracellular maspin.
Publications
-
Janice M. Burke, PhD, FARVO - in Memoriam (1946-2022).
(Carroll J, Besharse JC, Han DP, O'Brien WJ, Twining SS, Skumatz CM.) Exp Eye Res. 2022 Nov;224:109255 PMID: 36150545 SCOPUS ID: 2-s2.0-85150058281 09/24/2022
-
(Andreae EA, Warejcka DJ, Twining SS.) Mol Vis. 2020;26:540-562 PMID: 32818017 PMCID: PMC7406864 SCOPUS ID: 2-s2.0-85088944673 08/21/2020
-
(Andreae EA, Warejcka DJ, Twining SS.) Molecular Vision. 2020;26:540-562 SCOPUS ID: 2-s2.0-85088944673 01/01/2020
-
(Bohnsack RN, Warejcka DJ, Wang L, Gillespie SR, Bernstein AM, Twining SS, Dahms NM.) Invest Ophthalmol Vis Sci. 2014 Oct 30;55(12):7697-708 PMID: 25358730 PMCID: PMC4249949 SCOPUS ID: 2-s2.0-84914145905 11/02/2014
-
(Barnes J, Warejcka D, Simpliciano J, Twining S, Steet R.) J Biol Chem. 2012 Mar 02;287(10):7526-34 PMID: 22262853 PMCID: PMC3293579 SCOPUS ID: 2-s2.0-84857750167 01/21/2012
-
(Wang L, Pedroja BS, Meyers EE, Garcia AL, Twining SS, Bernstein AM.) PLoS One. 2012;7(3):e33915 PMID: 22470492 PMCID: PMC3309951 SCOPUS ID: 2-s2.0-84863358460 04/04/2012
-
(Warejcka DJ, Narayan M, Twining SS.) Exp Eye Res. 2011 Nov;93(5):618-27 PMID: 21810423 PMCID: PMC3221939 SCOPUS ID: 2-s2.0-81355161428 08/04/2011
-
(Endsley MP, Hu Y, Deng Y, He X, Warejcka DJ, Twining SS, Gonias SL, Zhang M.) J Biol Chem. 2011 Jul 15;286(28):24599-607 PMID: 21606500 PMCID: PMC3137035 SCOPUS ID: 2-s2.0-79960129686 05/25/2011
-
Identification of phosphorylation sites on extracellular corneal epithelial cell maspin.
(Narayan M, Mirza SP, Twining SS.) Proteomics. 2011 Apr;11(8):1382-90 PMID: 21365746 PMCID: PMC3098045 SCOPUS ID: 2-s2.0-79953706279 03/03/2011
-
PepD participates in the mycobacterial stress response mediated through MprAB and SigE.
(White MJ, He H, Penoske RM, Twining SS, Zahrt TC.) J Bacteriol. 2010 Mar;192(6):1498-510 PMID: 20061478 PMCID: PMC2832534 SCOPUS ID: 2-s2.0-77949371118 01/12/2010
-
Residues essential for plasminogen binding by the cation-independent mannose 6-phosphate receptor.
(Bohnsack RN, Patel M, Olson LJ, Twining SS, Dahms NM.) Biochemistry. 2010 Jan 26;49(3):635-44 PMID: 20028034 PMCID: PMC2814163 SCOPUS ID: 2-s2.0-74949084342 12/24/2009
-
(Narayan M, Twining S.) Exp Eye Res. 2010 Jan;90(1):2-3 PMID: 19615364 PMCID: PMC2789168 SCOPUS ID: 2-s2.0-70749148972 07/21/2009

