
Michaela Patterson, PhD
Associate Professor
Locations
- Cell Biology, Neurobiology and Anatomy
MEB M4575
Cardiovascular Research Center
Contact Information
Education
PhD, University of California, Los Angeles, CA
BS, Bates College, Lewiston, ME
Research Interests
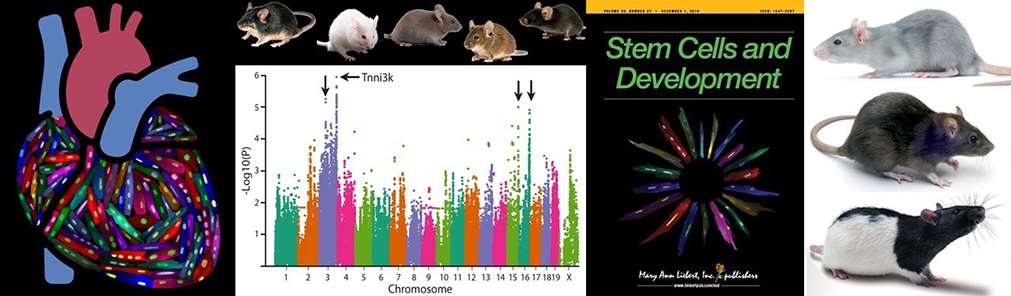
Our lab’s scientific interests lie at the intersection of cellular ploidy, genetic diversity, basal heart physiology, and tissue repair. We employ a mix of forward genetics approaches and resources to identify novel players in the traits we are exploring, along with classic reverse genetics and engineered animal models to assess causative relationships between identified genes and correlated phenotypes. Some specific areas of interest include:
1) Validation of the genetic underpinnings of cardiomyocyte ploidy and heart regeneration.
Patterson et al., Nature Genetics 2017 examined the relationship between cardiomyocyte ploidy and myocardial regeneration. Using a collection of 120 inbred mouse strains known as the Hybrid Mouse Diversity Panel, we uncovered multiple genetic loci associated with cardiomyocyte ploidy. We went on to validate the causative gene from the first locus, which proved to be Tnni3k.
The Patterson Lab has multiple projects underway (along with others still looking for a project lead) surrounding the other loci to arise from this screen. Using a variety of reverse genetic tools, including genetically engineered mice, viral induction, and more, some of the questions we commonly ask are:
- Is a candidate gene in the locus responsible for cardiomyocyte ploidy phenotypes AND what role do they play post-MI?
- Do the candidate genes have “functional” variants and what effect do they have on the gene’s molecular function and on ploidy?
- How do multiple genes, which individually influence ploidy, act together?
- Not all candidate genes function in cardiomyocytes, but instead act indirectly through another cell. What other cell types influence cardiomyocyte ploidy and MI outcomes?
2) New genetic screens to identify more players in cardiomyocyte polyploidization and outcomes after heart attack.
Our past work utilized the Hybrid Mouse Diversity Panel, a collection of inbred mouse strains designed to support genome-wide association studies. Our colleagues here at the Medical College of Wisconsin have recently rederived the rat equivalent resource - the Hybrid Rat Diversity Panel. The rat panel has many benefits that make it a superior genetic resource to the mouse panel, most notably that it is four times more genetically diverse. We have multiple projects underway, many of them collaborative, to examine a multitude of phenotypes across the Rat Panel. Phenotypes range from assessment of relevant cell populations to basal heart physiology to outcomes after heart attack and more. This arm of the Patterson lab has endless possibilities for expansion.
3) Polyploidy in other cell types.
Cardiomyocytes are not the only somatic cell in the body that displays substantial polyploidy. By examining the frequency of polyploid cells in other tissues we hope to better understand the overarching function of polyploidy in cell biology and determine if cell types utilize overlapping or distinct molecular mechanisms to drive polyploidization.
4) Bring your own ideas!
We are actively recruiting trainees at the graduate student and postdoctoral levels. We welcome new ideas to transform the way we think about tissue regeneration an repair. If you are interested in joining or rotating in the Patterson Lab, please contact Michaela Patterson, PhD, at mpatterson@mcw.edu.
Publications
-
(Akins KA, Arkatkar A, Flinn MA, Sandor N, Purdy AL, Brusacoram AL, Swift SK, Buday S, Choudhury P, Cui M, Lin BL, O'Meara CC, Patterson M.) JACC Basic Transl Sci. 2026 Feb;11(2):101455 PMID: 41534459 PMCID: PMC12830150 01/15/2026
-
(Gural B, Kirkland L, Hockett A, Sandroni P, Zhang J, Rosa-Garrido M, Swift SK, Chapski DJ, Flinn MA, O'Meara CC, Vondriska TM, Patterson M, Jensen BC, Rau CD.) PLoS Genet. 2025 Jul;21(7):e1011807 PMID: 40705823 PMCID: PMC12309993 SCOPUS ID: 2-s2.0-105011521361 07/24/2025
-
Protocol for quantifying murine cardiomyocyte cell division by single-cell suspension.
(Swift SK, Purdy AL, Patterson M.) STAR Protoc. 2024 Dec 20;5(4):103452 PMID: 39520685 PMCID: PMC11585686 11/13/2024
-
(Swift SK, Purdy AL, Buddell T, Lovett JJ, Chanjeevaram SV, Arkatkar A, O'Meara CC, Patterson M.) Cell Rep Methods. 2024 Sep 16;4(9):100860 PMID: 39255794 PMCID: PMC11440799 SCOPUS ID: 2-s2.0-85204511356 09/11/2024
-
(Gural B, Kirkland L, Hockett A, Sandroni P, Zhang J, Rosa-Garrido M, Swift SK, Chapski D, Flinn MA, O'Meara CC, Vondriska TM, Patterson M, Jensen BC, Rau CD.) bioRxiv. 2024 Aug 10 PMID: 39149394 PMCID: PMC11326268 08/16/2024
-
Runx1 is sufficient but not required for cardiomyocyte cell-cycle activation.
(Akins KA, Flinn MA, Swift SK, Chanjeevaram SV, Purdy AL, Buddell T, Kolell ME, Andresen KG, Paddock S, Buday SL, Veldman MB, O'Meara CC, Patterson M.) Am J Physiol Heart Circ Physiol. 2024 Aug 01;327(2):H377-H389 PMID: 38847758 PMCID: PMC11442100 SCOPUS ID: 2-s2.0-85199812831 06/07/2024
-
Global IL4Rα blockade exacerbates heart failure after an ischemic event in mice and humans.
(Alvarez-Argote S, Almeida VA, Knas MC, Buday SL, Patterson M, O'Meara CC.) Am J Physiol Heart Circ Physiol. 2024 May 01;326(5):H1080-H1093 PMID: 38426866 PMCID: PMC11380969 SCOPUS ID: 2-s2.0-85190400498 03/01/2024
-
IL-13 promotes functional recovery after myocardial infarction via direct signaling to macrophages.
(Alvarez-Argote S, Paddock SJ, Flinn MA, Moreno CW, Knas MC, Almeida VA, Buday SL, Bakhshian Nik A, Patterson M, Chen YG, Lin CW, O'Meara CC.) JCI Insight. 2024 Jan 23;9(2) PMID: 38051583 PMCID: PMC10906228 SCOPUS ID: 2-s2.0-85183312687 12/06/2023
-
The genetics of cardiomyocyte polyploidy.
(Buddell T, Purdy AL, Patterson M.) Curr Top Dev Biol. 2024;156:245-295 PMID: 38556425 04/01/2024
-
Tnni3k influences cardiomyocyte S-phase activity and proliferation.
(Purdy AL, Swift SK, Sucov HM, Patterson M.) J Mol Cell Cardiol. 2023 Oct;183:22-26 PMID: 37597489 PMCID: PMC11645536 08/20/2023
-
Cardiomyocyte ploidy is dynamic during postnatal development and varies across genetic backgrounds.
(Swift SK, Purdy AL, Kolell ME, Andresen KG, Lahue C, Buddell T, Akins KA, Rau CD, O'Meara CC, Patterson M.) Development. 2023 Apr 01;150(7) PMID: 36912240 PMCID: PMC10113957 SCOPUS ID: 2-s2.0-85152488769 03/14/2023
-
Myofibroblast Ccn3 is regulated by Yap and Wwtr1 and contributes to adverse cardiac outcomes.
(Flinn MA, Alvarez-Argote S, Knas MC, Almeida VA, Paddock SJ, Zhou X, Buddell T, Jamal A, Taylor R, Liu P, Drnevich J, Patterson M, Link BA, O'Meara CC.) Front Cardiovasc Med. 2023;10:1142612 PMID: 36998974 PMCID: PMC10043314 04/01/2023

