Pui-Ying (Penny) Lam, PhD
Assistant Professor
Locations
- Neuroscience Research Center
Contact Information
Education
PhD, University of Wisconsin-Madison
MPhil, The Hong Kong University of Science and Technology
BSc (Hons), The Hong Kong University of Science and Technology
Biography
Dr. Lam received her doctoral training at the University of Wisconsin-Madison in Dr. Anna Huttenlocher’s laboratory and postdoctoral training from Harvard Medical School and The University of Utah in Dr. Randall Peterson's laboratory. Her laboratory is broadly interested in small molecule tool development as well as the use of these tools along with in vivo imaging and modern molecular approaches to interrogate biological processes in health and disease. Dr. Lam received an American Heart Association postdoctoral fellowship, as well as a K99/R00 NIH BRAIN Initiative Advanced Postdoctoral Career Transition Award.
We are hiring
We are recruiting highly motivated trainees at all levels. If you are interested in joining or rotating in the Lam Lab, please contact Pui-Ying Lam, PhD at plam@mcw.edu. We are also hiring research technologists.
Research Interests
Disease Models & Mechanisms Journal Cover
December 2022; Volume 15, No. 12
Develop novel chemo-optogenetic tools for in vivo applications
Optogenetic/chemo-optogenetic tools accelerate basic research by providing the necessary means for manipulating cell activities using light with high temporal and spatial specificity. Our lab performs small molecule screens to discover chemo-optogenetic actuators to address some of the limitations of currently available optogenetic tools. We developed a zebrafish behavioral screening assay to identify photoactivatable compounds for endogenous vertebrate targets. As an example of our discoveries, our recent work published in JACS (Lam et al., 2020) described TRPswitch, a novel tunable chemo-optogenetic system based on a high-conductance cation channel, TRPA1, coupled to photo-activatable channel ligands (Figure 1). The new chemo-optogenetic tools we develop will benefit a wide range of researchers and have the potential to open up new areas of research. This project is supported by an NIH grant from the Brain initiative (R00NS112599).
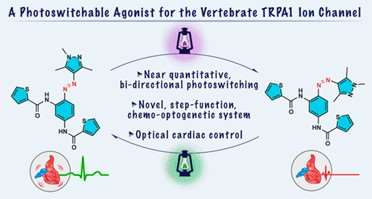
Figure 1. TRPswitch—A Step-Function Chemo-optogenetic Ligand for the Vertebrate TRPA1 Channel. Lam et al., JACS (2020)
Inflammation in the central nervous system
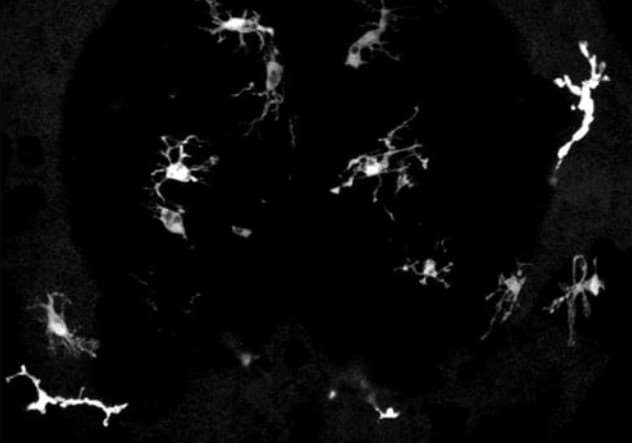
Uncontrolled neuroinflammation is recognized as a hallmark of neurological disease. Microglia, the resident macrophages of the brain, are highly susceptible to subtle changes in their environment. It is important to understand the behavior and regulation of immune cells in the intact brain, as well as to identify modulators that ameliorate pathogenesis of neurological diseases. To understand innate immunity and neuroinflammation in vivo, my group utilizes a synergistic approach that includes live imaging of transparent vertebrate fish models (zebrafish and Danionella cerebrum), optogenetic manipulation, in vivo small molecule screening, as well as novel chemo-optogenetic tool development. We aim to unravel how inflammation, particularly neuroinflammation, is regulated and to identify agents that provide beneficial modulation of immune function. Our work will offer new insights into innate immunity, provide novel tools to study and control innate immune cell function, and ultimately provide novel therapeutic options to improve human health.
Movie 1. In vivo confocal time-lapse movie showing microglia behavior and morphology in a live larval zebrafish brain.
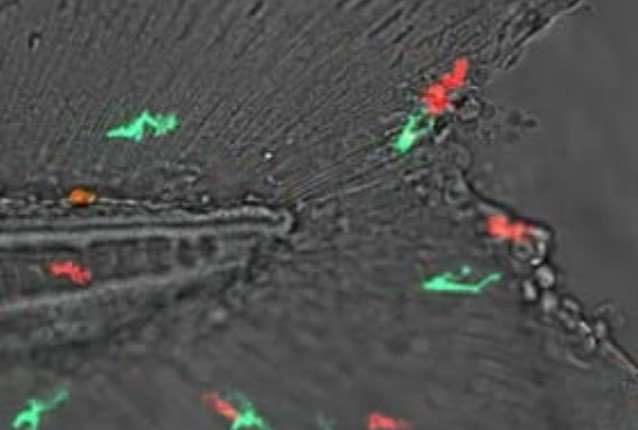
Movie 2. Neutrophils (red) and macrophages (green) respond to a tail fin wound in larval zebrafish.
Publications
-
The conducting state of TRPA1 modulates channel lateral mobility
(Sampieri A, Padilla-Flores T, Thawani AR, Lam PY, Fuchter MJ, Peterson R, Vaca L.) Cell Calcium. December 2023;116 SCOPUS ID: 2-s2.0-85172678285 12/01/2023
-
Longitudinal in vivo imaging of adult Danionella cerebrum using standard confocal microscopy.
(Lam PY.) Dis Model Mech. 2022 Dec 01;15(12) PMID: 36398624 PMCID: PMC9844135 SCOPUS ID: 2-s2.0-85144499718 11/19/2022
-
TRPswitch-A Step-Function Chemo-optogenetic Ligand for the Vertebrate TRPA1 Channel.
(Lam PY, Thawani AR, Balderas E, White AJP, Chaudhuri D, Fuchter MJ, Peterson RT.) J Am Chem Soc. 2020 Oct 14;142(41):17457-17468 PMID: 32966062 PMCID: PMC8011302 SCOPUS ID: 2-s2.0-85092945882 09/24/2020
-
(Houseright RA, Rosowski EE, Lam PY, Tauzin SJM, Mulvaney O, Dewey CN, Huttenlocher A.) Sci Rep. 2020 Sep 24;10(1):15716 PMID: 32973200 PMCID: PMC7518243 SCOPUS ID: 2-s2.0-85091432160 09/26/2020
-
Cyp1 Inhibition Prevents Doxorubicin-Induced Cardiomyopathy in a Zebrafish Heart-Failure Model.
(Lam PY, Kutchukian P, Anand R, Imbriglio J, Andrews C, Padilla H, Vohra A, Lane S, Parker DL Jr, Cornella Taracido I, Johns DG, Beerens M, MacRae CA, Caldwell JP, Sorota S, Asnani A, Peterson RT.) Chembiochem. 2020 Jul 01;21(13):1905-1910 PMID: 32003101 PMCID: PMC7500981 SCOPUS ID: 2-s2.0-85080951472 02/01/2020
-
Boholamide A, an APD-Class, Hypoxia-Selective Cyclodepsipeptide.
(Torres JP, Lin Z, Fenton DS, Leavitt LU, Niu C, Lam PY, Robes JM, Peterson RT, Concepcion GP, Haygood MG, Olivera BM, Schmidt EW.) J Nat Prod. 2020 Apr 24;83(4):1249-1257 PMID: 32186874 PMCID: PMC10172148 SCOPUS ID: 2-s2.0-85082040097 03/19/2020
-
(Fischer RS, Lam PY, Huttenlocher A, Waterman CM.) Dev Biol. 2019 Jul 01;451(1):86-95 PMID: 30193787 PMCID: PMC7082808 SCOPUS ID: 2-s2.0-85053325520 09/09/2018
-
Developing zebrafish disease models for in vivo small molecule screens.
(Lam PY, Peterson RT.) Curr Opin Chem Biol. 2019 Jun;50:37-44 PMID: 30928773 PMCID: PMC6800242 SCOPUS ID: 2-s2.0-85063391592 04/01/2019
-
Efficient Front-Rear Coupling in Neutrophil Chemotaxis by Dynamic Myosin II Localization.
(Tsai TY, Collins SR, Chan CK, Hadjitheodorou A, Lam PY, Lou SS, Yang HW, Jorgensen J, Ellett F, Irimia D, Davidson MW, Fischer RS, Huttenlocher A, Meyer T, Ferrell JE Jr, Theriot JA.) Dev Cell. 2019 Apr 22;49(2):189-205.e6 PMID: 31014479 PMCID: PMC6708378 SCOPUS ID: 2-s2.0-85064090810 04/25/2019
-
Noncanonical translation via deadenylated 3' UTRs maintains primordial germ cells.
(Jin YN, Schlueter PJ, Jurisch-Yaksi N, Lam PY, Jin S, Hwang WY, Yeh JJ, Yoshigi M, Ong SE, Schenone M, Hartigan CR, Carr SA, Peterson RT.) Nat Chem Biol. 2018 Sep;14(9):844-852 PMID: 29988067 PMCID: PMC6800240 SCOPUS ID: 2-s2.0-85049618175 07/11/2018
-
(Barros-Becker F, Lam PY, Fisher R, Huttenlocher A.) J Cell Sci. 2017 Nov 15;130(22):3801-3808 PMID: 28972134 PMCID: PMC5702045 SCOPUS ID: 2-s2.0-85034080622 10/04/2017
-
A high-conductance chemo-optogenetic system based on the vertebrate channel Trpa1b.
(Lam PY, Mendu SK, Mills RW, Zheng B, Padilla H, Milan DJ, Desai BN, Peterson RT.) Sci Rep. 2017 Sep 19;7(1):11839 PMID: 28928472 PMCID: PMC5605526 SCOPUS ID: 2-s2.0-85029827979 09/21/2017