
Jenifer Coburn, PhD
Professor, Medicine (Infectious Diseases) and Microbiology & Immunology; Center for Infectious Disease Research
Locations
- Center for Infectious Disease Research
TBRC C3980
Contact Information
General Interests
Education
Research Experience
- Borrelia Infections
- Spirochaetales Infections
- Tick-Borne Diseases
Research Interests

Research interests focus on pathogenic spirochetes, a group of bacteria that are able to cause persistent, disseminated infections in immunocompetent animals, including humans. We are currently working with Borrelia burgdorferi, which is maintained in a tick-animal cycle in nature. We also work with another pathogenic spirochete, Leptospira interrogans. Leptospires are maintained in infected animals in nature, but can also survive in water and mud. The focus of our work with both Borrelia and Leptospira is to identify and then test the biologic significance of bacterial proteins that help the bacteria bind to mammalian cell surface receptors, to identify the mammalian cell surface receptors recognized by the bacteria, and ultimately the biological and pathologic significance of the bacterial-mammalian receptor interaction.
In the Borrelia work, we have two main projects ongoing in the lab. In one, we are trying to understand the mechanisms behind the requirement for the B. burgdorferi protein, P66, for the bacteria to cause infection in mammals. P66 binds to mammalian cell surface receptors called integrins and serves as a porin in the bacterial outer membrane. We know that the integrin binding function is important for the bacteria to cross endothelial layers and disseminate to different sites in the body. In another Borrelia project, we developed a new experimental model to determine the roles of bacterial adhesive proteins in how the bacteria colonize different tissues in mammals, and how they survive the mammalian defenses in the bloodstream.

Figure 1. This image is an Ixodes scapularis tick. Shown is the nymphal stage of the tick; the scale bar is 1 mm so these ticks are tiny. These ticks are important in transmitting Borrelia burgdorferi to new host animals, and sometimes to humans.
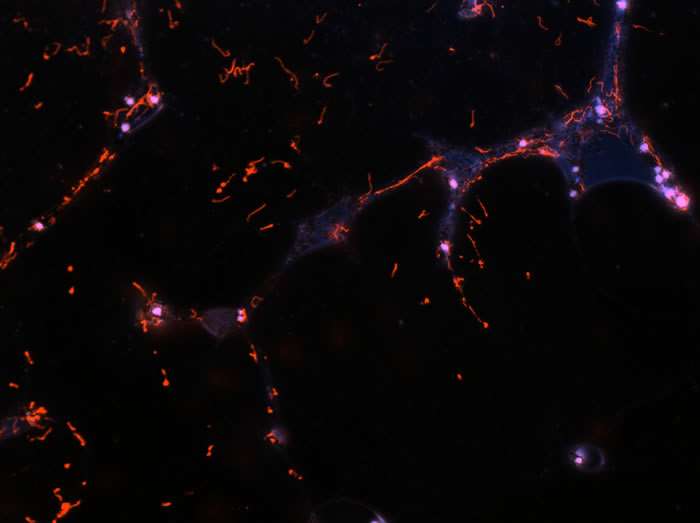
Figure 2: B. burgdorferi producing a red fluorescent protein in a dispersed tick midgut.
In the Leptospira work, we also focus in how the bacteria interact with endothelial cells. In severe cases of leptospirosis, widespread endothelial damage is seen, and this is associated with hemorrhage. L. interrogans binds to an endothelial cell surface receptor called VE-cadherin, which helps the endothelial cells form cell-cell junctions that maintain the integrity of small blood vessels. We are currently determining how the bacteria disrupt cadherin-cadherin interactions, and determining whether the bacterial proteins that bind VE-cadherin are responsible for the endothelial disruption caused by the bacteria. In a second Leptospira project, we are working to identify the bacterial proteins that help the bacteria bind to kidney cells, as the kidneys are where the bacteria reside in a chronically infected animal and from where they are released into the environment.
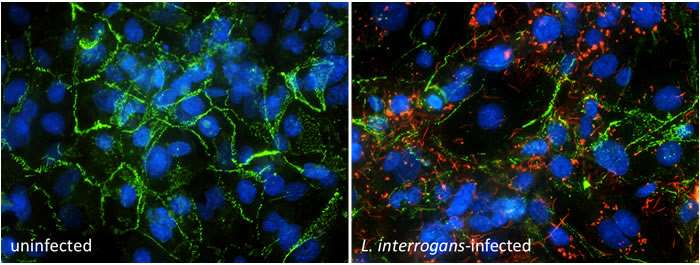
Figure 3: Leptospira interrogans (red) binding to human endothelial cells. An endothelial cell surface receptor for L. interrogans, VE-cadherin, is stained in green, but the cell-cell junctions are mostly disrupted. The endothelial cell nuclei are stained in blue.
Publications
-
In vitro and in vivo endothelial interactions of Leptospira species are markers of virulence.
(Anderson PN, Hahn BL, Thao G, Johnson MS, Giraud-Gatineau A, Gao Y, Picardeau M, Coburn J, Surdel MC.) PLoS Negl Trop Dis. 2026 Jan;20(1):e0013939 PMID: 41592120 PMCID: PMC12867331 SCOPUS ID: 2-s2.0-105029457041 01/27/2026
-
cGAS-STING dependent type I IFN reduces Leptospira interrogans renal colonization in mice.
(Gupta S, Matsunaga J, Ratitong B, Manion A, Ismaeel S, Valadares DG, West AP, Kerur N, Stehlik C, Dorfleutner A, Dagvadorj J, Coburn J, Wolf AJ, Morrissey MA, Cassel SL, Haake DA, Sutterwala FS.) PLoS Pathog. 2026 Jan;22(1):e1013250 PMID: 41499635 PMCID: PMC12795460 SCOPUS ID: 2-s2.0-105027301367 01/07/2026
-
cGAS-STING dependent type I IFN protects against Leptospira interrogans renal colonization in mice.
(Gupta S, Matsunaga J, Ratitong B, Ismaeel S, Valadares DG, West AP, Kerur N, Stehlik C, Dorfleutner A, Dagvadorj J, Coburn J, Wolf AJ, Cassel SL, Haake DA, Sutterwala FS.) bioRxiv. 2025 Jun 04 PMID: 40501870 PMCID: PMC12157660 06/12/2025
-
(Tal MC, Hansen PS, Ogasawara HA, Feng Q, Volk RF, Lee B, Casebeer SE, Blacker GS, Shoham M, Galloway SD, Sapiro AL, Hayes B, Torrez Dulgeroff LB, Raveh T, Pothineni VR, Potula HS, Rajadas J, Bastounis EE, Chou S, Robinson WH, Coburn J, Weissman IL, Zaro BW.) bioRxiv. 2024 Apr 30 PMID: 38746193 PMCID: PMC11092639 05/15/2024
-
Borrelia burgdorferi tolerates alteration to P66 porin function in a murine infectivity model.
(Fierros CH, Faucillion ML, Hahn BL, Anderson P, Bonde M, Kessler JR, Surdel MC, Crawford KS, Gao Y, Zhu J, Bergström S, Coburn J.) Front Cell Infect Microbiol. 2024;14:1528456 PMID: 39906208 PMCID: PMC11790652 SCOPUS ID: 2-s2.0-85216789061 02/05/2025
-
Leptospiral adhesins: from identification to future perspectives.
(Surdel MC, Coburn J.) Front Microbiol. 2024;15:1458655 PMID: 39206373 PMCID: PMC11350617 08/31/2024
-
(Tan X, Lin YP, Pereira MJ, Castellanos M, Hahn BL, Anderson P, Coburn J, Leong JM, Chaconas G.) PLoS Pathog. 2022 May;18(5):e1010511 PMID: 35605029 PMCID: PMC9166660 SCOPUS ID: 2-s2.0-85131457166 05/24/2022
-
(Curtis MW, Fierros CH, Hahn BL, Surdel MC, Kessler J, Anderson PN, Vandewalle-Capo M, Bonde M, Zhu J, Bergström S, Coburn J.) Front Cell Infect Microbiol. 2022;12:991689 PMID: 36211976 PMCID: PMC9539438 SCOPUS ID: 2-s2.0-85139437187 10/11/2022
-
(Surdel MC, Hahn BL, Anderson PN, Coburn J.) Front Cell Infect Microbiol. 2022;12:917963 PMID: 35937702 PMCID: PMC9354625 SCOPUS ID: 2-s2.0-85135274644 08/09/2022
-
(Surdel MC, Anderson PN, Hahn BL, Coburn J.) Front Cell Infect Microbiol. 2022;12:917962 PMID: 35923802 PMCID: PMC9339599 SCOPUS ID: 2-s2.0-85135250975 08/05/2022
-
Pathogenesis insights from an ancient and ubiquitous spirochete.
(Coburn J, Picardeau M, Woods CW, Veldman T, Haake DA.) PLoS Pathog. 2021 Oct;17(10):e1009836 PMID: 34673833 PMCID: PMC8530280 SCOPUS ID: 2-s2.0-85118294631 10/22/2021
-
(Coburn J, Garcia B, Hu LT, Jewett MW, Kraiczy P, Norris SJ, Skare J.) Curr Issues Mol Biol. 2021;42:473-518 PMID: 33353871 PMCID: PMC8046170 SCOPUS ID: 2-s2.0-85102523563 12/24/2020

