Evolving the Living Drug
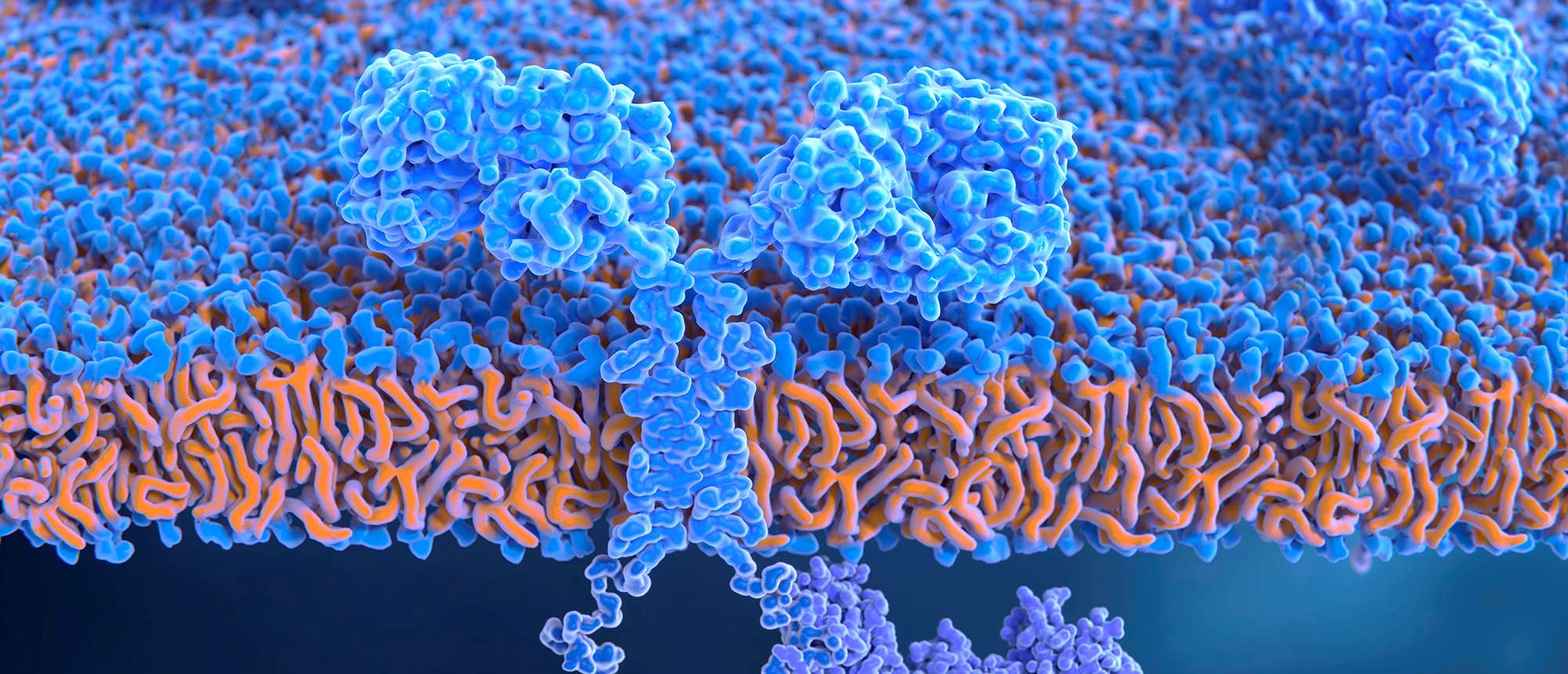
In CAR T-cell therapy, T cells isolated from a sample of a patient’s blood are genetically modified to contain CAR receptors on the cell surface (such as the one depicted here), which improve their ability to find and eliminate cancer cells.
The concept that the body’s immune system could be used to destroy cancer dates back at least 3,000 years to anecdotal accounts from ancient Egypt of tumors disappearing without treatment or after patients had recovered from infection. Developing and testing potential treatments based on bolstering a patient’s own immune system has required significant research to better understand the immune system and its role in cancer. A notable milestone was reached in the late 1960s with better insight gained about the T cell and its job eliminating virus-infected cells and cancer cells.
T cells recognize foreign entities by interacting with molecules known as antigens that are present on the surfaces of pathogens and cancer cells. Recent advances in genetic engineering allow clinicians and researchers to make the body’s own T cells even more effective against cancer cells through a technology known as chimeric antigen receptor (CAR) T-cell therapy.
“In this treatment approach, a sample of blood from a patient is separated into its components so that T cells can be isolated, genetically modified to contain CAR receptors on the cell surface, and then multiplied into the millions of cells needed for an effective dose that is injected back into the patient,” says Nirav Shah, MD, MSHP, MCW associate professor of medicine (hematology and oncology).
Some cancer cells can hide their surface antigens from naturally occurring T cells. CAR T cells, however, are better able to locate a specific antigen on the surface of cancer cells in order to find and eliminate them. Unlike chemotherapies and most other medicines (which are inanimate molecules that eventually break down within the body), CAR T cells are considered “living drugs” that can continue to expand while cancer cells with the specific target antigen are present in the body.
Currently, long-term progression-free survival after treatment with current FDA-approved CAR T-cell products ranges from 30-40 percent for aggressive B-cell non-Hodgkin lymphoma. The two approved therapies both target the cancer cell antigen CD19.
“Our number one goal is to continue to evolve CAR T-cell therapy and improve outcomes through innovation,” Dr. Shah notes. He and his team recently published results in Nature Medicine regarding a first-in-the-world phase I clinical trial that simultaneously targets two different cancer cell antigens – CD19 and CD20 – in individuals suffering from relapsed and refractory B-cell non-Hodgkin lymphomas.
“We know that cancer cells in patients who have relapsed after receiving current CAR T-cell therapies demonstrate less of the CD19 antigen as an adaptation. The goal of this trial was to demonstrate the safety and potential efficacy of an approach targeting two antigens as a solution to this challenge,” Dr. Shah explains.
Within 28 days of the treatment, 82 percent of the 22 study participants who achieved the target dose had responded positively to the treatment. Six months later, more than half were in complete remission, which means that the signs and symptoms of cancer were undetectable.
“We’ve now demonstrated safety and feasibility and learned a lot about the best dosage. I’m encouraged by the results and look forward to learning more about the effectiveness of the treatment in the next trial with a larger number of participants,” Dr. Shah adds. He will be the principal investigator of a national, multicenter phase II trial that may start enrolling participants in the first quarter of 2021.
– Greg Calhoun



