Microhelix Paves Way for Low-Cost Benchtop X-band Pulse EPR Spectrometers
 Electron paramagnetic resonance (EPR) experiments using double electron–electron resonance (DEER) spectroscopy monitor conformational changes and interactions between biomacromolecules in different environments and can complement techniques such as nuclear magnetic resonance, X-ray diffraction, and cryogenic electron microscopy. However, the adoption of DEER spectroscopy, and pulse EPR in general, is limited by the high-powered, high-cost microwave amplifiers required to supply the microwave power needed to excite the sample.
Electron paramagnetic resonance (EPR) experiments using double electron–electron resonance (DEER) spectroscopy monitor conformational changes and interactions between biomacromolecules in different environments and can complement techniques such as nuclear magnetic resonance, X-ray diffraction, and cryogenic electron microscopy. However, the adoption of DEER spectroscopy, and pulse EPR in general, is limited by the high-powered, high-cost microwave amplifiers required to supply the microwave power needed to excite the sample.
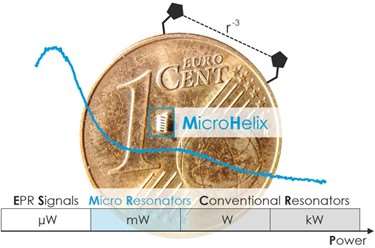
In their article titled “Milliwatt Three- and Four-Pulse Double Electron Electron Resonance for Protein Structure Determination” and published in Physical Chemistry Chemical Physics, authors Jason Sidabras, PhD, MCW assistant professor of biophysics, and Markus Teucher, PhD, and Alexander Schnegg, PhD, of the EPR Research Group at the Max Planck Institute for Chemical Energy Conversion in Germany, explore the use of a self-resonant microhelix at X-band (nominally 9.5 GHz; pictured) to obtain DEER data using three orders of magnitude less power (43 mW compared with 45 W) than commercially available resonators. The experiments detailed in the article were performed with as little as 85 nl of sample, facilitating microfluidic sample exchange.
The authors assert that eliminating the power requirements paves the way for an affordable benchtop pulse EPR spectrometer for protein structure determination that can be made widely available to the scientific community.

