Novel CAR-T Cell Immunotherapy for Lymphoma Advances to Phase II Clinical Trial
Study Results Published, Result of Collaborative Effort Led by Medical College of Wisconsin Researchers
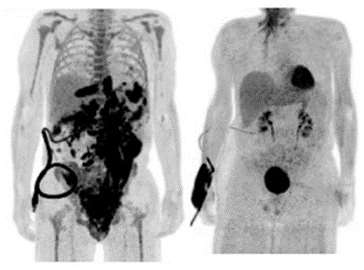
Milwaukee, Oct. 5, 2020 – A novel cancer therapy studied and developed at the Medical College of Wisconsin (MCW) with promising clinical outcomes is leading to a larger phase II trial to improve on the current standard of care. Results of phase I of the first-in-the-world double targeted chimeric antigen receptor (CAR) T-cell therapy clinical trial were published in Nature Medicine today.
This is a novel, cell-based treatment against cancer targeting two proteins (antigens CD19 and CD20) on the surface of cancer cells. This CAR-T cell therapy trial began in October 2017 and resulted in safe and promising outcomes for patients with relapsed and refractory B cell non-Hodgkin lymphomas which are cancers of the immune system. MCW researchers collected patients’ own immune cells (T-cells) and then used a specially engineered virus to augment their ability to identify and kill cancerous cells and effectively destroy the lymphoma. While phase I focused on safety and feasibility of the treatment, a multi-institutional phase II is being developed to determine the true efficacy and understand how the nuances of the treatment process can result in excellent outcomes for a larger subset of patients.
All patients in the clinical trial had failed prior treatments and their cancer had relapsed. Within 28 days of the CAR-T cell therapy, 82 percent responded positively. Six months later, more than half of the patients’ cancer remained in remission. A higher dose of the treatment correlated with a prolonged remission, a trend the researchers plan to study further in the trial’s second phase.
“Immuno-oncology using T-cell treatments shows incredible promise for cancer patients,” said Nirav Shah, MD, MSHP, principal investigator of the trial, associate professor of medicine in the division of Hematology and Oncology at MCW, and lymphoma and stem cell transplant specialist at the Froedtert & the Medical College of Wisconsin Clinical Cancer Center at Froedtert Hospital. “This is a giant leap forward in personalized medicine. Very few cancer centers offer these novel combination of resources and high level of personalized medicine expertise. The encouraging results of this trial pave the way for more effective and efficient treatment options.”
While CAR-T cell therapy has been under development since 2012, the Froedtert & MCW Clinical Cancer Center treated the first patients using this dual-targeted gene therapy. The new treatment genetically alters a person’s own immune cells to target cancer cells in a unique and personalized fashion, a significant departure from more routine chemotherapy.
The cell product used for treatment was manufactured using the CliniMACS Prodigy® device, which is part of an automated CAR-T cell manufacturing platform developed by Miltenyi Biotec, a global company dedicated to enabling cellular and gene therapies. A collaboration of scientists and clinicians recognized the potential of this treatment several years ago and began acquiring necessary equipment to prepare for a clinical trial. Housed at the Froedtert & MCW Clinical Cancer Center, the CliniMACS Prodigy® device enabled the research team to conduct the CAR-T cell immunotherapy through a self-contained, desktop system, producing new cells ready to be infused back into a patient’s bloodstream within 14 days. With the device, the entire process was performed locally at Froedtert Hospital, saving precious time for the patients.
The launch of this clinical trial is the result of decades of collaborative cancer and cellular immunotherapy research at the Blood and Marrow Transplant and Cellular Therapy program. Pioneers in the field of immunotherapy, these researchers worked on ideas of harnessing the body’s own immune system to fight cancer cells, leading to innovative alternatives to traditional chemotherapy, radiation and transplants, that are often effective in killing cancer cells but also frequently damage the body’s healthy cells. This knowledge paved the way for the CAR-T cell treatment, which trains the patient’s own immune cells to kill the cancer, rather than relying on foreign, toxic substances.
“We are harnessing knowledge from decades of research to improve outcomes for patients with lymphoma,” said Parameswaran Hari, MD, MRCP, MS, senior author and professor and chief of the Division of Hematology and Oncology at MCW, specializing in treating individuals with myeloma, leukemia and lymphoma at the Froedtert & MCW Clinical Cancer Center. “There is amazing potential here for the future of not just lymphoma patients but patients with many other cancers.”
This research was made possible through philanthropic dollars raised by the Children’s Wisconsin Foundation and the MACC Fund (Midwest Athletes Against Childhood Cancer, Inc.) and their support of the Cell Therapy Lab at MCW. Lentigen Technology, a Miltenyi Biotec company, provided the lentiviral (LV20.19) vector used in this study and partial funding support for data collection. The Drive for the Cure Foundation and the Froedtert & MCW Cancer Center provided additional partial funding.
“This clinical trial demonstrates the strength of the partnerships across this campus and the power of academic medicine,” said David Margolis, MD, program director of the Bone Marrow Transplant (BMT) and Cellular Therapy Program and professor of pediatrics and interim chair of the Department of Pediatrics at MCW. “The expertise of MCW researchers, state-of-the-art facilities of Children’s and Froedtert and generosity of this community made this work possible.”
Keep up with the latest news. Sign up for Newsroom Alerts.
Latest press releases, stories and resources.
MCW Media Contacts
The media relations team at MCW is happy to assist in coordinating experts for interviews. Please reach out to us at:
media@mcw.edu
(414) 955-8764
Contact us


