Medical College of Wisconsin Cardiovascular Academic Initiative
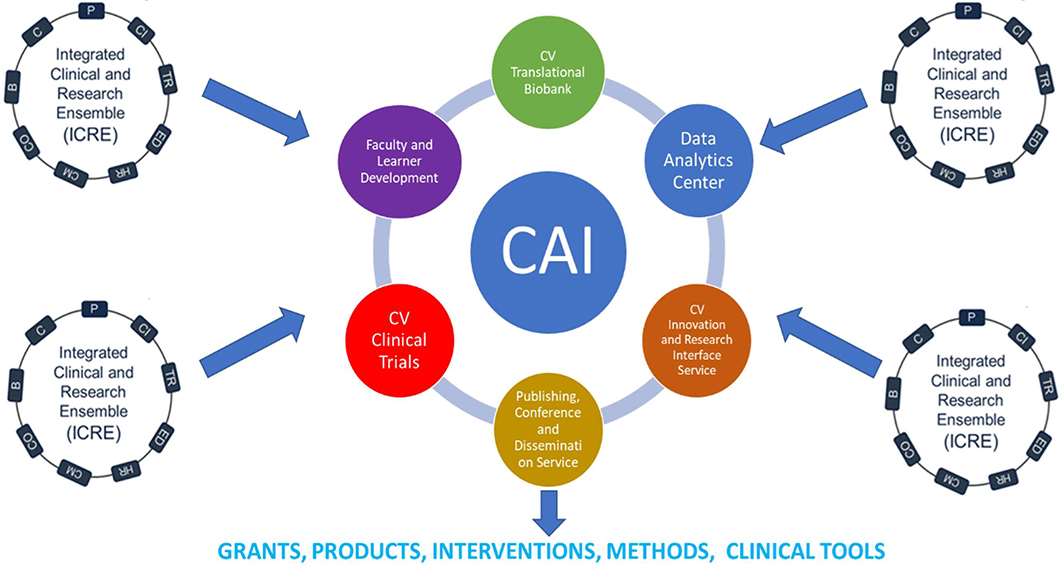
 The Cardiovascular Academic Initiative was formed in the fall of 2020 in response to the need(s) of building a more robust infrastructure for facilitating multi-directional cardiovascular translational research. The CAI builds upon MCW’s current strengths in the fundamental research in cardiovascular science research by leveraging existing assets available through the Clinical and Translational Research Initiative (CTSI) of Southeast Wisconsin and MCW’s Cardiovascular Research Center to support building a robust, synergistic translational research infrastructure to speed the translation of cardiovascular research to the bedside as well as find novel solutions to critical 21st century problems in cardiovascular care.
The Cardiovascular Academic Initiative was formed in the fall of 2020 in response to the need(s) of building a more robust infrastructure for facilitating multi-directional cardiovascular translational research. The CAI builds upon MCW’s current strengths in the fundamental research in cardiovascular science research by leveraging existing assets available through the Clinical and Translational Research Initiative (CTSI) of Southeast Wisconsin and MCW’s Cardiovascular Research Center to support building a robust, synergistic translational research infrastructure to speed the translation of cardiovascular research to the bedside as well as find novel solutions to critical 21st century problems in cardiovascular care.
Our Goals
- Create collaborative spaces for innovation in cardiovascular research and clinical care
- Facilitate the translation of cardiovascular research between basic sciences, early human studies, and clinical practice
- Train the next generation of leaders in cardiovascular research and education
Meet Our Team

Michael E. Widlansky, MD, MPH
Northwestern Mutual Professor of Cardiovascular Medicine, Department of Medicine and Pharmacology; Director of the Cardiovascular Research Center

Michelle Knezevich
Data Analyst

Monica Driessen
Clinical Research Coordinator

Nicholas Durst
Clinical Research Assistant II
Funding Opportunities
Patient-Oriented Cardiovascular Academic Projects (PCAP)
- Application for up to $10,000 will be considered (one year of funding)
- Derived from funds via the 2018/2019 Heart of the Matter fundraiser
- Project must meet the NIH’s definition of Patient-Oriented Research
- Cannot supplant current funding
- Reviewed on a rolling basis over the 2023-24 academic year
- RFA: PCAP (PDF) and Non-Supplanting Attestation (PDF).
Cardiovascular Focused Ensembles
- In collaboration with the Clinical and Translational Science Institute (CTSI) of Southeast Wisconsin of Southeast Wisconsin, the CAI will be issuing a request for applications (RFA) for the formation of cardiovascular-focused research ensembles.
- Successfully formed Ensembles selected for this program are eligible for an initial $50,000 line of credit to support their approach. In addition, the Ensemble will receive administrative support from the CTSI as well as clinical research coordinator and data analytics support from the CAI.
- Applications for the Cardiovascular Focused Ensemble formation for 2023 are now closed. Stay tuned for 2024 information.
- The CAI has funded the following:
- Funded May 1, 2022
- Dr. Marcie Berger & Dr. Stacey Gardiner – entitled "CTSI Community Care Initiative Risk & Prevention Program Ensemble"
- Dr. Kevin Cohoon – entitled "Rapid, Accurate Diagnosis of Traumatic Brain Injury/Concussion at the Point of Injury Ensemble"
- Funded June 1, 2023
- Dr. Rashimi Sood – entitled "Prediction of Time to Delivery Ensemble"
- Funded May 1, 2022
CAI Services
Contact Us
The Cardiovascular Academic Initiative is currently conducting drug and/or device trials in the cardiac subspecialties listed. However, this does not limit what we may be working on tomorrow.
Please contact the Cardiovascular Academic Initiative directly by emailing CardioAcademicInitiative@mcw.edu.
Medical College of Wisconsin
The Hub for Collaborative Medicine
CAI, 5th Floor
8701 Watertown Plank Rd.
Milwaukee, WI 53226
Program Director
Michael Widlansky, MD, MPH